SURFACE DEALING
- Share

Surface Treatment
Surface treatment is a process for manually forming a surface layer on the surface of a substrate material that is different from the mechanical, physical, and chemical properties of the substrate. The purpose of surface treatment is to meet the corrosion resistance, wear resistance, decoration or other special functional requirements of the product. For metal castings, our common surface treatment methods are mechanical grinding, chemical treatment, surface heat treatment, spray surface, surface treatment is to clean, clean, deburr, degrease, deoxidize the surface of the workpiece.
Normal Hardware Surface Treatment

Mirror Polished
Mechanical mirror polishing is a smooth, shiny mirror-like surface on a metal material through a buffing process (rough grinding, fine grinding) and a polishing process (WENDT three-step polishing).
The purpose of polishing is to obtain a smooth and smooth surface. At this time, there is a very fine and uniform wear scar on the grinding surface. Polishing is divided into rough grinding and fine grinding.
The polishing process
1. Rough grinding Rough grinding is the modification of rough surfaces and irregular shapes.
2. Fine grinding After rough grinding, the metal surface still has deep wear marks, which need to be eliminated in the fine grinding to prepare for polishing.
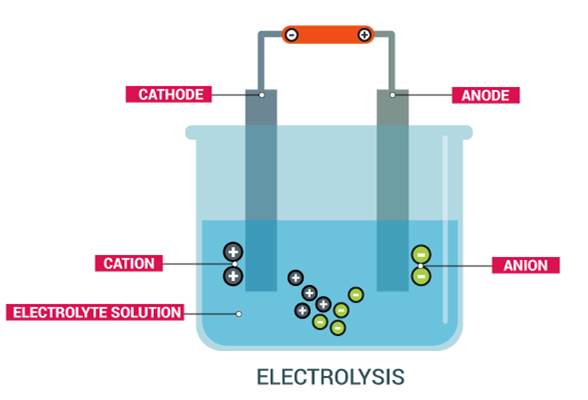
Electrolysis
Electrolysis is a process in which an electric current is passed through an electrolyte solution or a molten electrolyte (also referred to as an electrolyte) to cause a redox reaction on a cathode and an anode, and an electrochemical cell may undergo an electrolysis process when a direct current voltage is applied.
Electrolysis is a process of synthesizing a high-purity substance for chemical synthesis and a surface treatment of a material by an electrochemical reaction occurring at an interface between an electrode as an electron conductor and an electrolyte as an ion conductor. When energized, the cations in the electrolyte move toward the cathode, absorb electrons, and reduce to form new substances; the anions in the electrolyte move toward the anode, emit electrons, generate oxidation, and generate new substances. For example, electrolytically smelting sodium chloride.

Hot-Dip Galv
Hot-dip galvanizing is the reaction of a molten metal with an iron matrix to produce an alloy layer, thereby combining the substrate and the plating. Hot-dip galvanizing is the first step of pickling steel parts in order to remove iron oxide on the surface of steel parts. After pickling, it is cleaned by ammonium chloride or zinc chloride aqueous solution or mixed solution of ammonium chloride and zinc chloride. And then sent to the hot dip plating tank. Hot-dip galvanizing has the advantages of uniform plating, strong adhesion and long service life.
Phosphorization
Phosphorization is a chemical and electrochemical reaction to form a phosphate chemical conversion film. The phosphate conversion film formed is called a phosphate film. The purpose of phosphating is mainly to provide protection to the base metal, to prevent the metal from being corroded to a certain extent; to base the paint before painting, to improve the adhesion and corrosion resistance of the paint layer; to reduce friction in the metal cold working process Lubrication use.
